 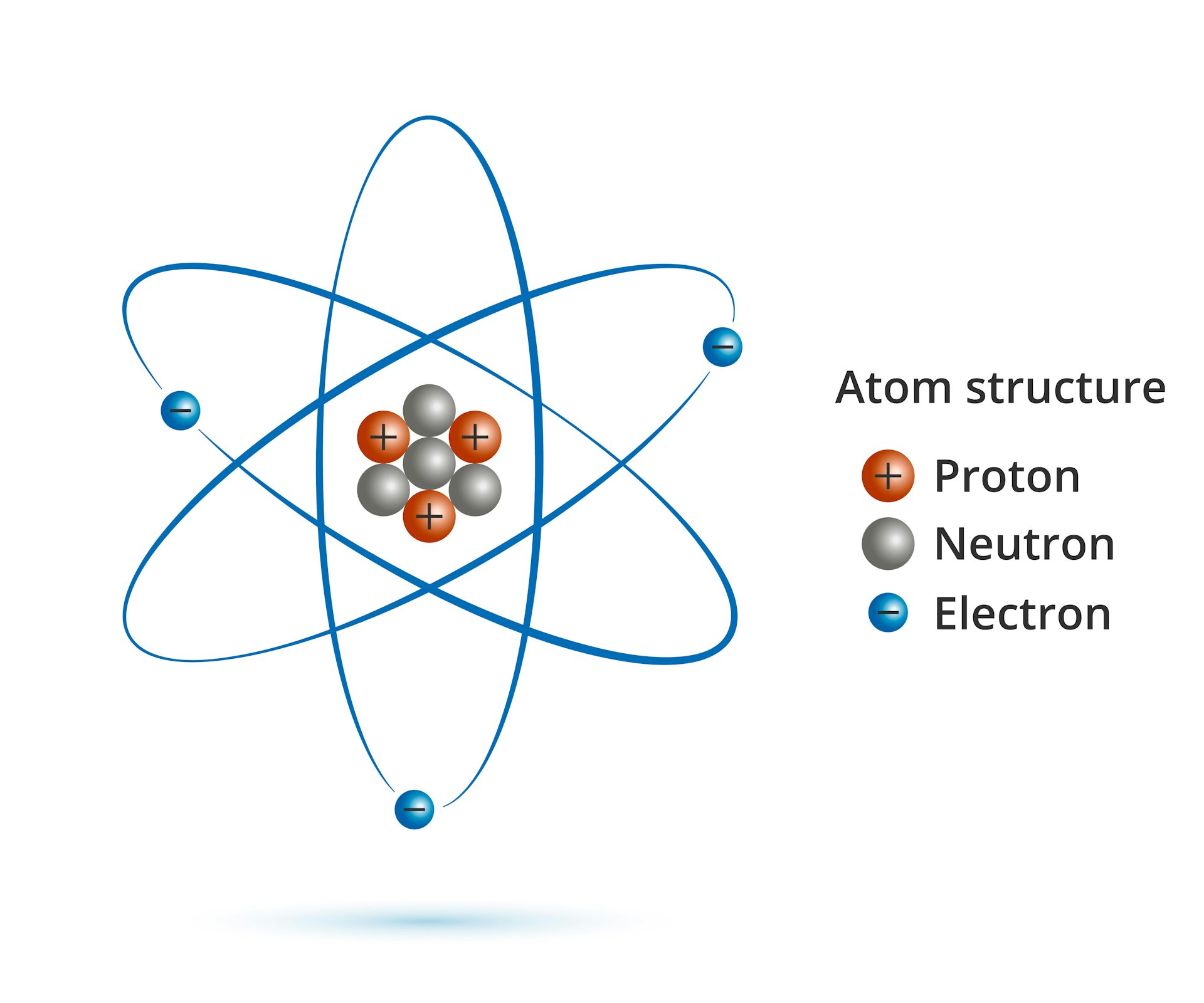 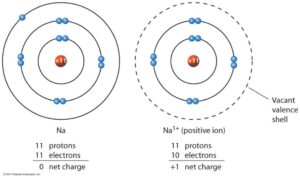
Neutrons = 45-21=24 (it is the same isotope) Electrons = 21-3 = 18 (a 3+ ion means 3 electrons have been lost) What is the number of protons, neutrons and electrons in an Sc 3+ ion of the same isotope? Protons = 21 (it is still scandium) What is the number of protons, neutrons and electrons in an atom of scandium-45? Protons = 21 (look it up on periodic table) Neutrons = 45-21 = 24 (given mass number - number of protons) Electrons = 21 (equal to number of protons for an atom) An ion with a negative charge has gained electrons – again the magnitude of the charge indicates how many electrons have been gained. The magnitude of the charge indicates the number of electrons that have been lost. An ion with a positive charge has lost electrons (there are now more protons than electrons). While atoms do not lose or gain protons or neutrons, they can lose or gain electrons to become ions with an overall charge. Therefore number of neutrons = mass number – atomic numberīecause an atom has no overall charge, there must be exactly the same number of electrons as protons in an atom. The total number of protons and neutrons (nucleons) in an atom is the mass number. The periodic table is arranged in order of increasing atomic number – each different type of atom is identified by its unique number of protons. The number of protons in an atom is the atomic number. Numbers of protons, neutrons and electrons Most of an atom is empty space – protons, neutrons and electrons are very small compared to atoms, but because protons and neutrons weigh much more than electrons, virtually all the mass in an atom is in the nucleus. The nucleus (containing the protons & neutrons) has an overall positive charge, and the electrons orbiting around it are negatively charged, so an atom has charge distribution – positive in the middle and negative surrounding this region. The particles from which atoms are made are protons, neutrons and electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
